System Suitability Testing is used in pharmaceutical labs to ensure that the lab’s systems (such as chromatography systems and spectrophotometers) are functioning properly and providing reliable results. This is done by performing measurements on Quality Control (QC) samples: blanks (water) or reference standards. The QC samples must pass predefined acceptance criteria in order for the system to be considered suitable for use. Veeva LIMS leverages Test Definition Variations to allow Lab Analysts to easily perform System Suitability Testing alongside our standard Test Execution process. This feature must be configured by an Admin before it can be used.
Design Data Admin Steps
In order for Lab Analysts to be able to create QC samples and complete System Suitability Testing, a Design Data Admin must first set up a Test Definition Variation for each Test Definition that requires System Suitability Testing. In addition to the standard Test Definition Variation setup steps, the Design Data Admin should ensure the following:
- The Test Definition Variation has the correct value selected in the Quality Control Sample Type field. The default values available are Blank and Reference Standard, but a Vault Admin can also configure custom values.
- The Test Definition Criteria is defined.
About Test Definition Criteria
Test Definition Criteria is a child object of Test Definition that tracks the criteria that Quality Control Samples should be tested against. Conceptually, Test Definition Criteria is the same as Spec Data Criteria, except that it is associated with a Test Definition instead of Spec Data. Because Quality Control Samples are created ad hoc and therefore are not associated with any Spec Data, they require a different solution to define their criteria.
Note: Test Definition Criteria uses the same Criteria Evaluation object as Spec Data Criteria. The Criteria Evaluation*_ object has reference fields for both, but only one should be used at a time. If a given _Criteria Evaluation record has both of those fields populated, an error will occur.
Creating Test Definition Criteria
Before creating Test Definition Criteria, you must set up a Test Definition Variation with the required Quality Control Sample Type. To create Test Definition Criteria:
- Navigate to the Test Definition for which you’ve created your Test Definition Variation.
- Expand the Test Definition Criteria section and click Create.
- Select Quality Control Sample Criteria.
- Click Continue.
- Select the Test Definition Variation you created.
- Select a Result to Evaluate.
- Select a Variation Result to Evaluate.
- Select an Indication. This determines how the Test Definition Criteria is represented during Test Execution (if applicable) and whether failing this criteria prevents spec conformance.
- Enter a Criteria formula.
- Enter a Criteria Description. This functions as a more human-readable representation of the Criteria formula.
- Click Save.
Performing System Suitability Testing
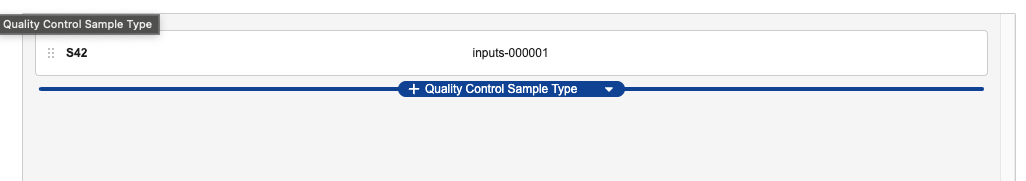
When configured by an Admin, Lab Analysts can create Quality Control Samples as needed from the Edit Test Set dialog in the Test Execution UI. To do so, complete the following steps:
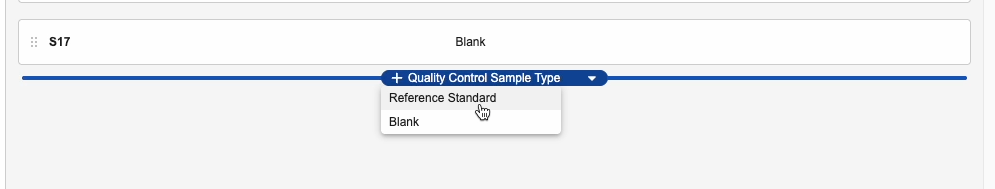
- In the Edit Test Set dialog, hover your mouse directly below the list of currently included samples and click the + Quality Control Sample Type button that appears.
- Select a Sample Type in the drop-down menu that appears.
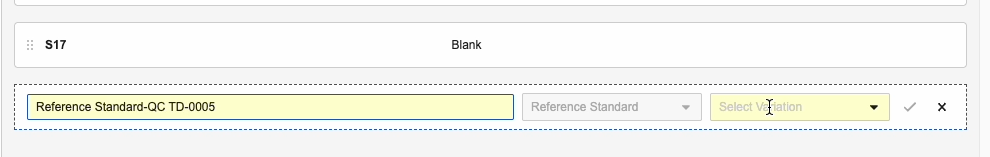
- Optional: Edit the auto-populated Name.
- Select a Variation.
- Click Save.
Once the Lab Analyst saves a Quality Control Sample, Veeva LIMS checks for an existing Quality Control Spec Execution for the current Test Set and, if one does not already exist, creates one automatically. After that, the system creates a new Lab Test associated with the Spec Execution for each newly created Quality Control Sample. These Lab Tests are all based on the selected Lab Test Definition Variation. Next, the system creates any necessary child objects for the Lab Tests, such as Test Steps, Test Results, Test Inputs, Test Result Variables, and Criteria Evaluations.
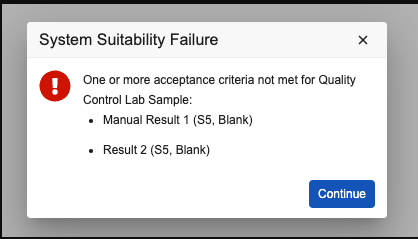
Flow When a Quality Control Sample Does Not Meet its Criteria
When a Quality Control Sample does not meet its criteria, this means that the System Suitability Test has failed; that is, the system is not delivering accurate results. In this case, after clicking the Evaluate, Next, or Verify button, a dialog appears informing the Lab Analyst of which QC results failed conformance and they must choose how to proceed.
When the Lab Analyst clicks the Continue button, the Test Set Invalidate workflow begins. Depending on how the workflow has been configured, the Analyst can choose whether to continue testing or invalidate the Test Set. The recommended workflow configuration moves the Test Set to the In Review state and gives the Analyst these three options:
- Continue Testing: The Test Set moves back to the In Progress state and the Analyst can continue testing.
- Invalidate Test Set and Retest: The Test Set moves to the Rejected state and the Retest action runs on all existing Tests in the Test Set.
- Invalidate Test Set, Resampling Required: The Test Set moves to the Rejected state.
Relevant Permissions
In order to manage design data related to System Suitability Testing, Design Data Admins must have View permissions to the Lab Test Definition Version History control and Criteria control for the Lab Test Definition Criteria object as well as the Test Definition Criteria control for Spec Data object.
In order to perform data entry related to System Suitability Testing, Lab Analysts must have Edit access to the Invalid Reason and Invalid Category fields on the Test Set object.
Additionally, to interact with System Suitability Testing, all LIMS Users must have Read and Edit access to the Quality Control Sample object type on the Lab Sample object, Read access to the Quality Control Sample Criteria and Operation Criteria object types on the Lab Test Definition Criteria object, and Read access to the Quality Control Sample and Operational Criteria fields on the Spec Execution object.